Solar Light’s versatile SPF-290AS UV Transmittance / Sunscreen Protection Factor Analyzer is an integrated turnkey UV spectrophotometer, designed and optimized for the determination of SPF values on a variety of sunscreen and cosmetic products, as well as materials testing. This self-contained, attractive desktop unit reduces the need and associated cost for in-vivo testing. Covering both the UVB and UVA spectral regions, the system automatically scans from 290 to 400 nm, accumulating and storing data at intervals of 1, 2, or 5nm. The monochromatic protection factor (MPF) is determined for each of the selected wavelengths, and is used to calculate the SPF value, using solar irradiance and erythemal constants that are programmed into the software (but which can be easily modified.)
Compliant With:
ISO 23698:2024
ISO 23675:2024
ISO 24443
FDA
COLIPA
Boots Star Rating
Brochure
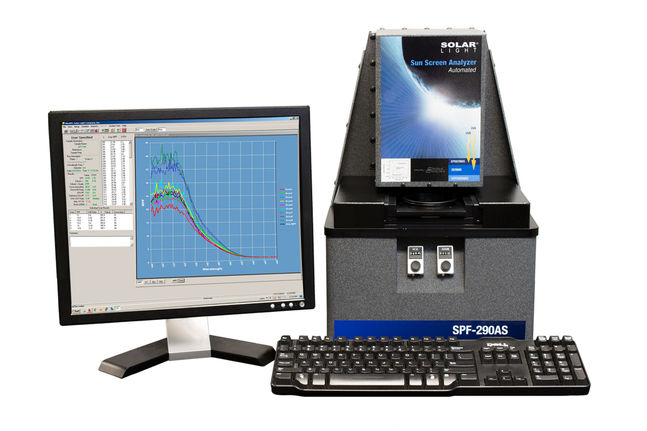
The SPF-290AS is the perfect choice for quick, efficient, and accurate analysis of:
Liquids
Lotions
Creams
Sprays
Gels
Powders
Emulsions
Textiles
Wavelength Range:
290 – 400 nm (range specified by international methods)
Wavelength Accuracy:
0.2% (0.58 nm – 0.80 nm)
Wavelength Reproducibility:
0.25 nm
Wavelength Interval:
1.00 nm
Absorbance:
0 – 3.2 A (Dual Doped PMMA Method)
SPF Measurement Range:
1 – 100+
Scan Time:
As little as 24 seconds
Wavelength Step Interval:
1 nm, 2 nm, or 5 nm. User selectable
Lamp:
Power stabilized Xenon 150W, 100% Ozone Free
Detector:
High Sensitivity Multialkali side-on PMT
Sample Positioning:
Automated X-Y Stage
Power Options:
110VAC-220VAC, 50/60 Hz
We are proud to present our range of featured products, each crafted to the highest standards of quality and performance.